Research
February 16, 2011
Emory a top contributor in drug discovery
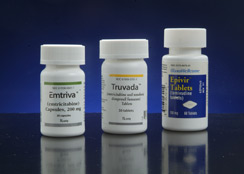
Emory-discovered HIV/AIDS drugs are taken by more than 94 percent of U.S. patients on therapy and by thousands more globally.
A new study finds that Emory University is the fourth largest contributor in the nation to the discovery of new drugs and vaccines by public-sector research institutions. The contributors include federally funded universities, research hospitals and federal laboratories.
The research was published in the Feb. 10 issue of the New England Journal of Medicine.
The study authors, from Boston University School of Management and a research foundation in Norway, identified 153 FDA-approved drugs and vaccines that were discovered at least in part by public-sector research institutions during the past 40 years. The top five contributors were the NIH (22), the University of California System (11), Memorial Sloan-Kettering (8), Emory University (7), and Yale University (6).
The seven Emory products included HIV/AIDS drugs lamivudine (3TC) and emtricitabine (FTC), discovered by Emory scientists Dennis Liotta and Raymond Schinazi and their former colleague Woo-Baeg Choi.
These two drugs are among the most commonly used and most successful HIV/AIDS drugs in the world, taken in some form by more than 94 percent of U.S. patients on therapy and by thousands more globally.
Start-ups and licensing are outgrowths
"We are extremely proud of our scientists and their lifesaving and life-enhancing discoveries," says President Jim Wagner. "This study illustrates once again that our nation's long-standing and world-leading policy of investment in research through universities and other public institutions, along with the responsible use of technology transfer, delivers a tremendous return through improved health for millions, innovative technologies, economic development and training for the next generation of innovators."
Over the past two decades, through its Office of Technology Transfer, Emory has launched 51 start-up companies and received more than $788 million in licensing revenues from drugs, diagnostics, devices and consumer products.
Currently, more than 50 products are in various stages of development or regulatory approval, with 27 having reached the marketplace and 12 more in human clinical trials. Emory continues to return the funds it receives from its technology transfer successes back into a variety of programs in research and science education.
